Background
Two formations in snow appeared in December 2009, one on December 19th and the other on December 22nd. (See http://bltresearch.com/robbert/snow09.php. Observations of them included:
a.) No footprints were found in the rings or elsewhere in the field;
b.) No marks of any kind in the centers of the circular rings;
c.) Rings were created by removal of the top 4 inches of snow, but snow was not removed down to the surface of the soil.
d.) No evidence of the snow that had been removed to create rings (where did it go?);
e.) No evidence of icing in the rings (which would be expected if something had been used to melt the snow in the rings).
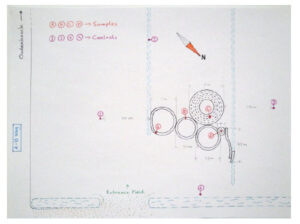
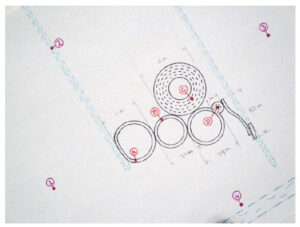
Another snow formation appeared on February 2, 2010. There was much less snow on the ground than in the above formations, and this one is also very “rough” looking, i.e. not as “geometric” or precise. And there are “scrape” marks, the same as appeared in the larger rings in the other two formations, as if somebody just dragged something around and around. There are no footprints anywhere in any of the rings, and even the slightest weight on the snow leaves a very clear mark. This time snow samples were collected for analysis. Following are diagrams of the formation with sampling locations, as well as photographs.
The objective of this analysis is to determine if there is any chemical signature which could indicate a substance placed on the field either prior to or following the discovery of the new snow formation.
Conclusions
- pH measurements and amounts of residues from the snow control and formation snows are equivalent. Ph measurements amounted to 5.8 for all samples. This is normal for snow. Residues amount (on average) to 0.1 g/l for all samples.
- No anomalous material is obvious in the formation snow samples. All snow samples (both control and formation) contain similar components, which can be considered as originating from the environment and some contamination. While they are qualitatively similar, they quantitatively vary in amounts of components. The control samples contain environmental materials such as inorganic sulfate and nitrate, humate and carboxylic acid salt functionalities, which resemble sodium formate (though this structure is not confirmed), and some natural ester. A trace amount of poly(dimethylsiloxane) contamination is also detected. This may be from the containers. The formation snows contain environmental components such as humate, and carboxylic acid salt similar to sodium formate, and inorganic nitrate and sulfate.
- It should be noted that inorganic nitrates appear to predominate only in snow formation samples C and D. The two nitrates appear to be different. Spectral bands in C are most similar to sodium nitrate and those in D are closer to ammonium nitrate. It is unknown if this is significant.
- No snow melting components were found in the formation samples. Sodium formate is possibly present, though not confirmed, in both control and formation
The Analysis
 Loading...
Loading...Follow The Black Vault on Social Media: